Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Shop Silylation
Silylation
Warning: Undefined array key "images" in /home/campbell/public_html/index.php(211) : eval()'d code on line 68
Warning: Trying to access array offset on value of type null in /home/campbell/public_html/index.php(211) : eval()'d code on line 68
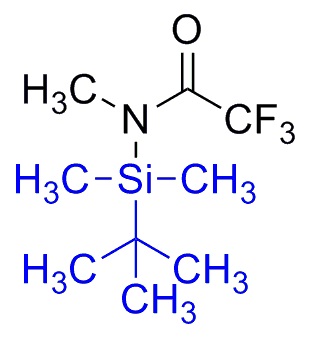
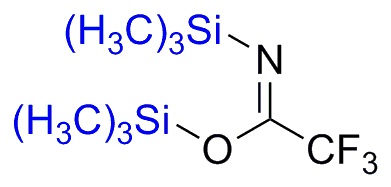
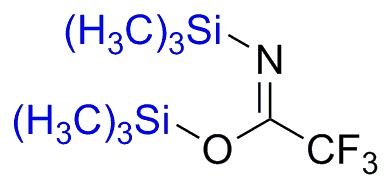
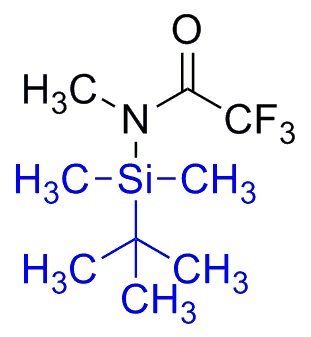
Silylation is the introduction of a substituted silyl group to a molecule. The process is the basis of organosilicon chemistry. Alcohols, carboxylic acids, amines, thiols, and phosphates can be silylated. The process involves the replacement of a proton with a trialkylsilyl group, typically trimethylsilyl (-SiMe3). Generally the substrate is deprotonated with a suitable strong base followed by treatment with a silyl chloride (e.g. trimethylsilyl chloride). Often strong bases such butyl lithium or a Grignard reagent are used, as illustrated by the synthesis of a trimethylsilyl ethers as protecting groups from an alcohol.
The introduction of a silyl group(s) gives derivatives of enhanced volatility, making the derivatives suitable for analysis by gas chromatography and electron-impact mass spectrometry (EI-MS). For EI-MS, the silyl derivatives give more favorable diagnostic fragmentation patterns of use in structure investigations, or characteristic ions of use in trace analyses employing selected ion monitoring and related techniques.
The introduction of a silyl group(s) gives derivatives of enhanced volatility, making the derivatives suitable for analysis by gas chromatography and electron-impact mass spectrometry (EI-MS). For EI-MS, the silyl derivatives give more favorable diagnostic fragmentation patterns of use in structure investigations, or characteristic ions of use in trace analyses employing selected ion monitoring and related techniques.
Warning: Undefined array key "product_controls" in /home/campbell/public_html/tool/marketplace/view/view.productCard.php on line 4
Warning: Undefined array key "product_controls" in /home/campbell/public_html/tool/marketplace/view/view.productCard.php on line 5
Deprecated: unserialize(): Passing null to parameter #1 ($data) of type string is deprecated in /home/campbell/public_html/tool/marketplace/view/view.productCard.php on line 5