Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Warning: Undefined array key "id" in /home/campbell/public_html/tool/marketplace/widget/marketSidebar.php on line 49
Shop Biotinylation Reagents
Biotinylation Reagents
Warning: Undefined array key "images" in /home/campbell/public_html/index.php(211) : eval()'d code on line 68
Warning: Trying to access array offset on value of type null in /home/campbell/public_html/index.php(211) : eval()'d code on line 68
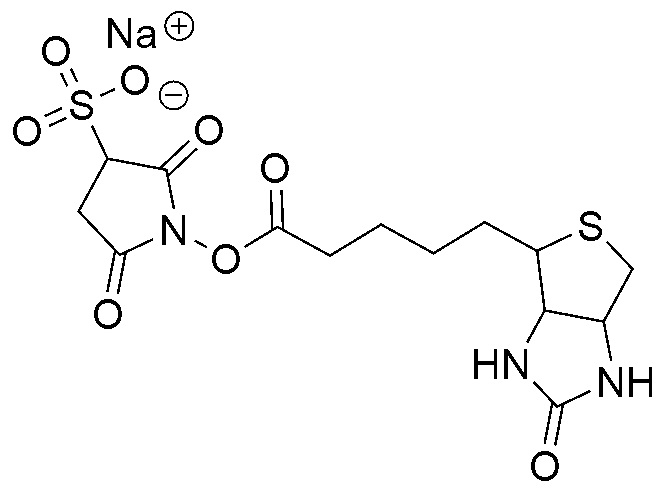
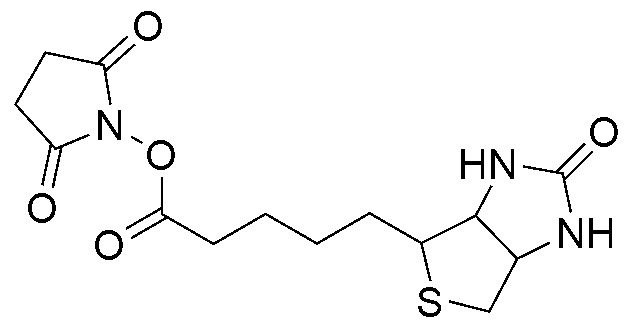
Biotinylation is the process of covalently attaching biotin to a protein, nucleic acid or other molecule. Biotinylation is rapid, specific and is unlikely to disturb the natural function of the molecule due to the small size of biotin (MW = 244.31 g/mol). Biotin binds to streptavidin and avidin with an extremely high affinity, fast on-rate, and high specificity, and these interactions are exploited in many areas of biotechnology to isolate biotinylated molecules of interest. Biotin-binding to streptavidin and avidin is resistant to extremes of heat, pH and proteolysis, making capture of biotinylated molecules possible in a wide variety of environments. Also, multiple biotin molecules can be conjugated to a protein of interest, which allows binding of multiple streptavidin, avidin or neutravidin protein molecules and increases the sensitivity of detection of the protein of interest. There is a large number of biotinylation reagents available that exploit the wide range of possible labelling methods. Due to the strong affinity between biotin and streptavidin, the purification of biotinylated proteins has been a widely used approach to identify protein-protein interactions and post-translational events such as ubiquitylation in molecular biology.